Storage condition assurance
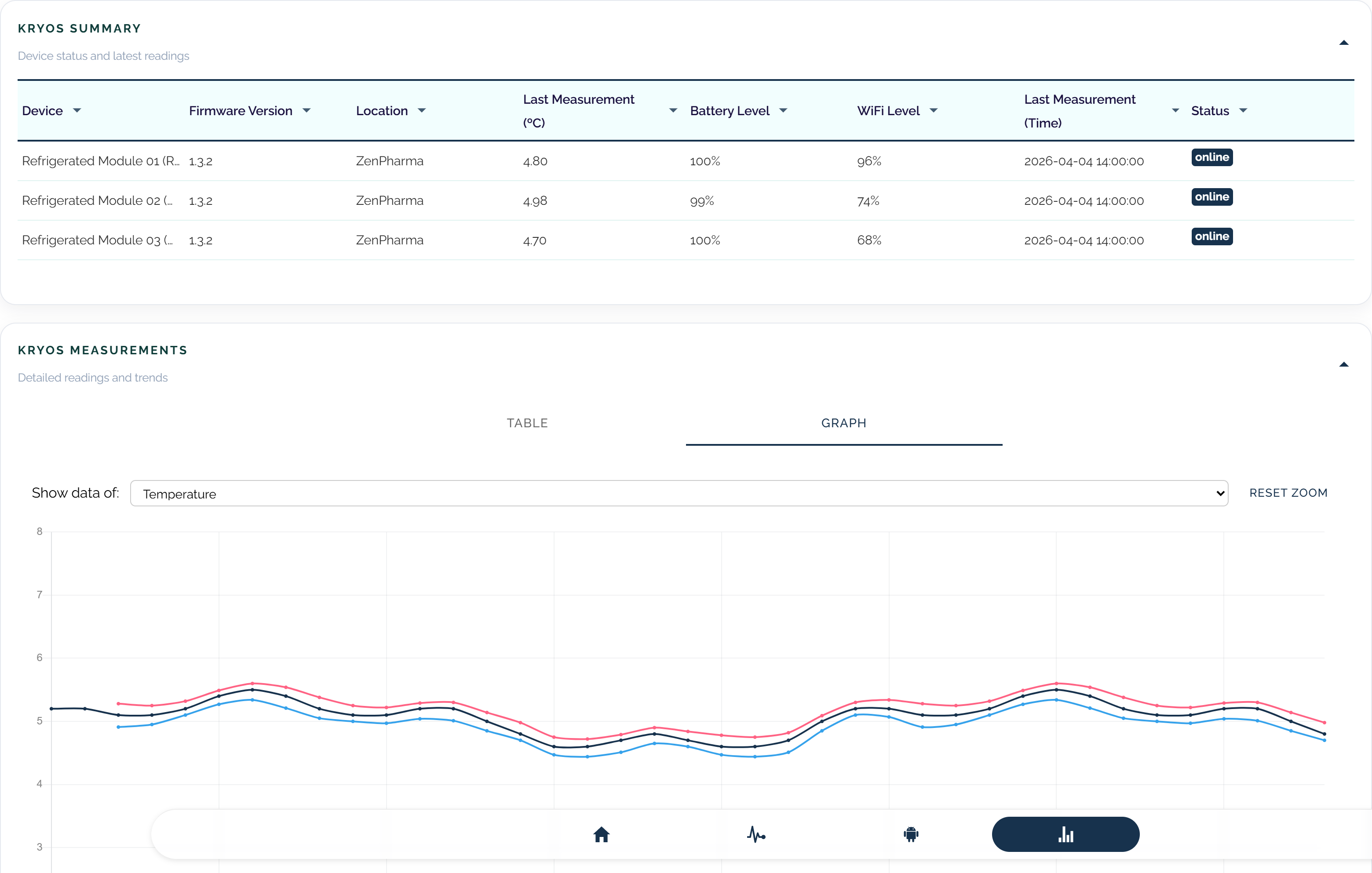
Monitor vaccine units continuously with threshold controls.
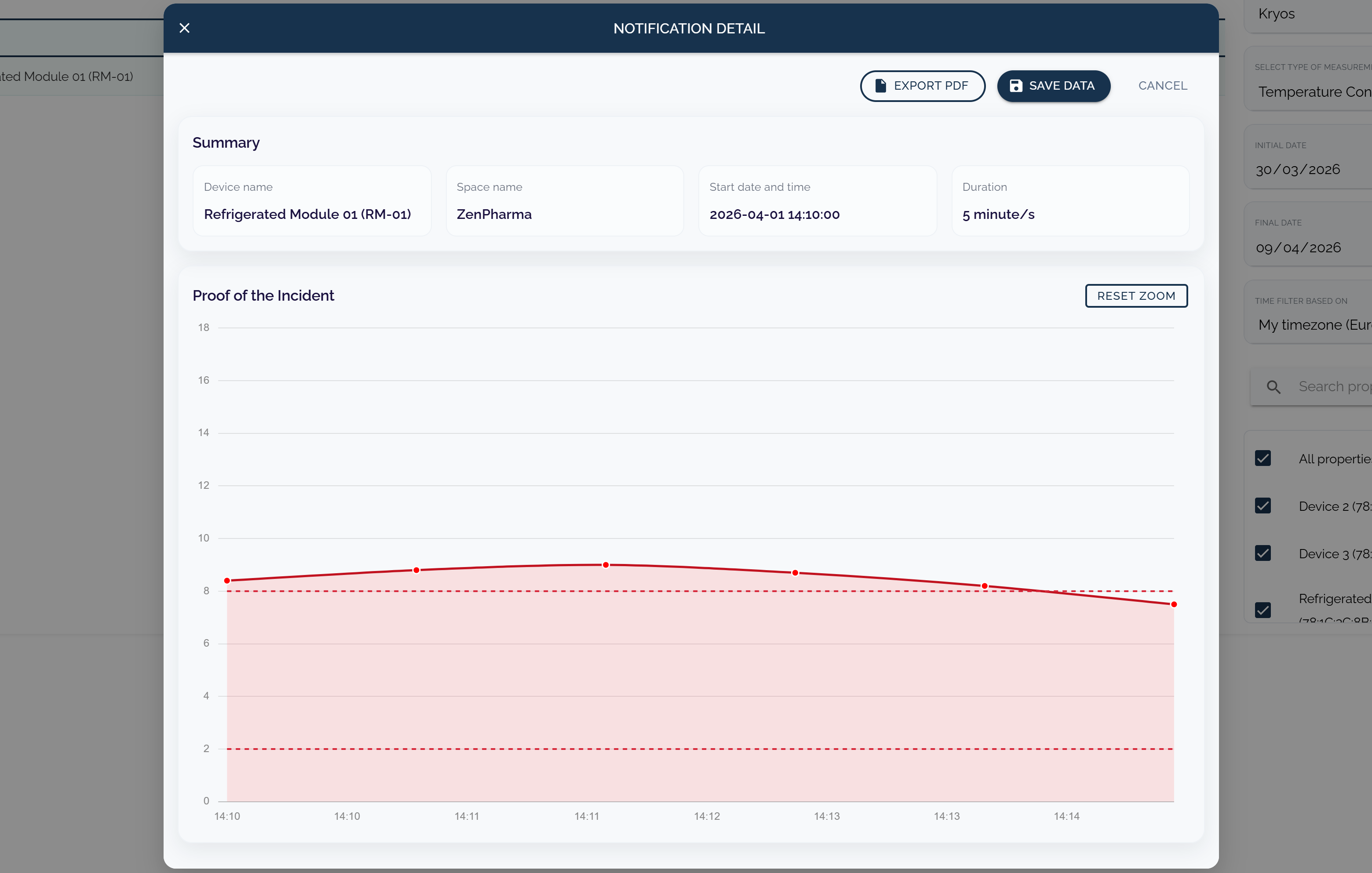
Maintain vaccine storage oversight with continuous readings, alerts, and structured incident timelines.
Monitor vaccine units continuously with threshold controls.
Capture how teams respond to storage deviations.
Preserve records for regulatory and internal review.
Deze pagina sluit aan op de kernbewijzen voor monitoring, respons en gedocumenteerde beoordeling.
Whether you monitor pharmacy refrigerators, medical storage, or transport conditions, we can help you build a cleaner and more reliable monitoring setup.
Kies een moment om uw workflow voor temperatuurmonitoring met KRYOS te bespreken: locaties, koelkasten, vriezers, ruimtes, routes, alarmen, rapporten, exports en implementatiebehoeften.